DARPP-32 Involvement in the Photic Pathway of the Circadian
Lily Yan,1 Jessica M. Bobula,1
Per Svenningsson,2 Paul Greengard,2 and Rae Silver1,3,4
1Department of Psychology, Columbia University, New York, New York 10027,
2Laboratory of Molecular and Cellular Neuroscience, The Rockefeller
University, New York, New York 10021, 3Department of Psychology,
Barnard College, New York, New York 10027, and 4Department of Anatomy
and Cell
Biology, College of Physicians and Surgeons, Columbia University, New York, New
York 10032
The multifunctional regulator of protein kinases and phosphatases dopamine- and
cAMP-regulated phosphoprotein of 32 kDa (DARPP-
32) is an important molecular target of the dopamine signaling pathway. In the
present study, we investigated the possible involvement
of DARPP-32 regulation in the circadian system using DARPP-32 knock-out (KO)
mice. These mice showed normal entrainment to a 12 h
light/dark cycle and free run in constant darkness with a period similar to that
of wild-type controls. After light exposure, however, the
behavioral phase-delay response and the expression of light-induced clock gene
mPer2 were attenuated in the DARPP-32 KO mice.
Attenuated phase delays were also seen in animals bearing a point mutation in
DARPP-32 at the PKA (Thr34) but not at the casein kinase
I (Ser130) phosphorylation site. We next examined DARPP-32 expression in the
retina and intergeniculate leaflet (IGL), both of which
convey light information to the suprachiasmatic nucleus (SCN), the locus of a
master circadian clock, and in theSCNitself. DARPP-32 was
expressed in the retina but not in the IGL or the SCN. The results indicate that
DARPP-32 is involved in the retinal pathway transmitting
photic information that resets the circadian clock.
Key words: DARPP-32; suprachiasmatic nucleus; phase shifts; circadian rhythm;
photic transmission; retina
Introduction
The suprachiasmatic nucleus (SCN) is the locus of a master clock
that organizes daily rhythms of physiology and behavior (Klein et
al., 1991). The SCN is synchronized to the local environment by
external cues, the most predictable of which is the daily light/dark
(LD) cycle. Phase-setting photic input is conveyed from the eye to
the SCN via a direct retinohypothalamic tract (RHT) and indirectly
by the geniculohypothalamic tract (GHT) from the intergeniculate
leaflet (IGL) (Moore and Lenn, 1972; Moore, 1982).
The present objective was to explore the possible role of
dopamine- and cAMP-regulated phosphoprotein of 32 kDa
(DARPP-32) in circadian rhythm generation and in providing
phase-setting information to the circadian system.
DARPP-32, a multifunctional regulator of protein kinases and
phosphatases, is an important molecular target of dopamine and
is expressed in neurons receiving dopaminergic input (Walaas et
al., 1983). DARPP-32 has, at least, four phosphorylation sites:
Thr34, Thr75, Ser97, and Ser130. The multiple functions of
DARPP-32 are achieved through phosphorylation at these sites.
Phosphorylation by protein kinase A (PKA) at Thr34 converts
DARPP-32 into a potent inhibitor of protein phosphatase-1
(PP-1) (Hemmings et al., 1984). The Ser97 and Ser130 sites can
be phosphorylated by casein kinases I and II, respectively, leading
to increased Thr34 phosphorylation (Girault et al., 1989; Desdouits
et al., 1995). Phosphorylation at the Thr75 site inhibits
PKA-induced phosphorylation at Thr34 (Bibb et al., 1999). The
activated or inhibited DARPP-32/PP-1 pathway regulates the activity
of a large array of downstream physiological effectors, including
neurotransmitter receptors, voltage-gated ion channels,
ion pumps, and transcriptional factors (Greengard, 2001; Svenningsson
et al., 2004).
Dopamine signaling has been implicated in circadian rhythm
regulation. Deficient light-masking response of dopamine D2 receptor
knock-out (KO) mice suggests that the dopamine pathway
is essential for light masking of circadian rhythms (Doi et al.,
2006). Dopaminergic signals may reach the circadian system
through a retinal or anSCNsite of action. In the retina, dopamine
plays an important role in light adaptation (Witkovsky, 2004),
and dopamine regulates the rhythmic expression of melanopsin,
a photopigment of intrinsically photosensitive retinal ganglion
cells (Sakamoto et al., 2005). In the SCN, dopaminergicD1 receptors
are present in both fetuses and adults (Weaver et al., 1992;
Ishida et al., 2002), but the phase-resetting effect of dopamine is
seen only prenatally (Viswanathan et al., 1994). Finally, in cultured
cell lines, dopamine signals enhance the transactivation
potentials of CLOCK:BMAL1, the primary positive regulator of
the molecular clock (Yujnovsky et al., 2006).
To examine the possible functional significance of DARPP-32
in circadian rhythmicity and photic entrainment, we compared
the responses of wild-type (WT), DARPP-32 KO (Fienberg and
Greengard, 2000), and phosphomutant DARPP-32 mice (Svenningsson
et al., 2003). The results indicate that DARPP-32 KO
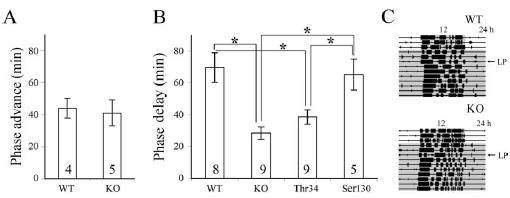
Figure 1. Phase shifting of wheel-running activity after
an advancing (CT 22) or delaying (CT 16) light pulse (300 lux for 30 min)
during subjective night. A, Phase-advance response. B, Phase-delay response
after 3 d in DD by WT, DARPP-32 KO, and mice
bearing a point mutation in Thr34 or Ser130. C, Actograms show phase-delay
response of representativeWT(top) or DARPP-32 KO
(bottom) mice after a light pulse (LP) on the third day of DD. Data are
presented as mean ±SEM. Numbers on the histogram
indicate the sample size. *p< 0.0125 (Bonferroni-corrected for multiple
comparisons). The gray area in C indicates the dark
phase.
mice have attenuated behavioral phase delays and
light-induced
expression of mPer2 mRNA in the SCN. Importantly, the results
show that DARPP-32 mRNA was expressed in the retina, but not
in the SCN or IGL. Our results provide the first evidence that
DARPP-32 is involved in the retinal pathway transmitting photic
signals to SCN.
Materials and Methods
Animals and housing. Male DARPP-32 KO mice, the Ser130 and Thr34
point mutants, and their WT littermate controls (C57BL/6 background)
were studied at ~5 weeks of age. All mice have been described previously
(Fienberg and Greengard, 2000; Svenningsson et al., 2003). Mice were
housed in a 12 h LD(300 lux) cycle for 2–4 weeks before being used in the
experiments. Food and water were available ad libitum. All experimental
procedures were approved by the Institutional Animal Care and Use
Committee of Columbia University.
For behavioral studies, mice were housed individually, and their
wheel-running behavior was monitored using DataQuest (Data Services,
St. Paul, MN); the data were analyzed using Clocklab (Actimetrics, Evanston,
IL). First, we entrained the WT and KO mice (n =10 per group) in
LD for 2 weeks and then transferred them to constant dark (DD) for
another 2 weeks to determine their free-running period. Next, we assigned
each genotype to two groups that were exposed to a light pulse
(300 lux for 30 min) at either circadian time 16 (CT 16) or CT 22. In the
next study, animals of four different genotypes (WT and KO, Ser130, and
Thr34 mutants; n =10 per group) were entrained to the LD cycle, transferred
to DD, and exposed to a light pulse at CT 16 on the third day in
DD, to assess their phase-delay response.
For mRNA analysis, DARPP-32 KO and WT animals (n =4 per
group) were exposed to a light pulse at CT 16 on the third day in DD and
killed 90 min later (CT 17.5). Control animals were killed at the same
time but did not receive light exposure.
In situ hybridization using digoxigenin-labeled cRNA probes. Mice were
deeply anesthetized (pentobarbital; 200 mg/kg) under a red safe light
( <1 lux) and perfused intracardially with 10 ml of saline and 20 ml of a
fixative (4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.4). The
brains were removed, postfixed, and cryoprotected for 48 h. To assess
light-induced mPer1 and mPer2 expression, serial coronal sections (40
m) were made from the rostral to the caudal end of the SCN using a
cryostat (Reichert-Jung, Heidelberg, Germany), and alternate sections
were collected for each probe. To investigate DARPP-32 expression in
the brain, 40 μm sections at the level of SCN and IGL were collected. The
IGL was defined precisely by processing alternate sections for NPY
immunostaining
(Takatsuji and Tohyama, 1989). To examine the retina,
the cornea and lens were removed from the eye, and the eyecups were
postfixed, cryoprotected, and sectioned at 40 μm.
The cRNA probes for mPer1 (nucleotide position 538 –1752), mPer2
(1– 638), and DARPP-32 (424 –910) were synthesized with a standard
protocol (Yan et al., 1999). In situ hybridization
was performed as described previously (Yan et
al., 1999; Yan and Silver, 2002). For each of
these transcripts, sense probes revealed no
staining.
Immunocytochemistry of NPY. For NPY immunocytochemistry
(ICC), sections were incubated
in the NPY antibody (1:10,000; generated
in rabbit; INCSTAR, Stillwater, MN) for 48 h at
4°C and then in Cy3 (cyanine 3)–donkey antirabbit
antibody (1:200; Jackson ImmunoResearch,
West Grove, PA) for 2 h at room temperature.
After the ICC reaction, sections were
coverslipped with Krystalon (EM Diagnostic
Systems, Gibbstown, NH).
Laser-capture microdissection of SCN tissue,
RNA extraction, and reverse transcription-PCR.
Fresh frozen forebrains were cut at 8 m, and
the sections containing SCN were mounted on
uncharged glass slides (VWR Scientific, West
Chester, PA). The sections were thawed at room
temperature and fixed in 75% ethanol. The sections were then stained
and dehydrated with HistoGene laser-capture microdissection (LCM)
frozen section staining kit (Arcturus Bioscience, Mountain View, CA).
LCM of SCN tissue was performed using a Pixcell II laser-capture microscope
(Arcturus Bioscience) with standard procedure (Emmert-Buck et
al., 1996). After visually identifying the SCN region, the SCN tissue was
lifted by a brief laser pulse and captured into a CapSure LCM cap (Arcturus
Bioscience). Approximately forty SCN sections from five animals
were pooled, and the total RNA was isolated using a PicoPure RNA
isolation kit (Arcturus Bioscience). cDNA was synthesized using a
RETROscript kit (Ambion, Austin, TX), and the quality of the cDNA was
evaluated by a cDNA integrity kit (Kirkegaard & Perry Laboratories,
Gaithersburg, MD). PCR was performed using a GeneAmp 2400 system
(PerkinElmer, Wellesley, MA).
Quantitative analysis. To assess mPer1 and mPer2 levels, images of
SCN were captured using a CCD video camera [Sony (Tokyo, Japan)
XC77] attached to a light microscope (BH-2; Olympus Optical, Tokyo,
Japan). Optical density (OD) was quantified using the NIH Image program
(version 1.61). The difference between SCN density and background
was used as theODvalue for each section. The averagedODvalue
of the sections from each brain was used as the OD value for one animal.
Results
DARPP-32 KO mice show attenuated phase delays
There were no significant differences in entrainment or freerunning
behavior between DARPP-32 KO mice and their WT
littermates, although they did differ in their phase-shifting behavior.
Under LD, the total running activity of the WT mice was
higher than that of the KO; however, the difference was not statistically
significant (32± 6 *103 vs 24 ±2 *103 wheel rotations/
d; p >0.05). Both WT and KO mice run predominantly
during the dark phase (95.9 ±1.3 vs 93.6± 1.6%; p > 0.05).
Under DD, the free-running period of the KO mice was about the
same as that of their WT littermates (23.84 ±0.14 vs 23.9 ±
0.16 h; p >0.05). We further examined the photic response of
these animals using a brief light exposure. After light exposure at
CT 22, there was no significant difference in the phase advances
between the DARPP-32 KO and WT mice (Fig. 1A). In contrast,
a light pulse at CT 16 revealed significant differences in the phasedelay
response between WT and DARPP-32 KO mice (Fig. 1B).
Comparison of WT, KO, and mutant animals with a point
mutation at either the Ser130 or the Thr34 phosphorylation site
of DARPP-32 indicated a similar phase-delay response in the
Ser130 mutant and WT mice, whereas the phase delay of the
Thr34 mutant was significantly less than WT but about the same
as that of the DARPP-32 KO mice (Fig. 1B).
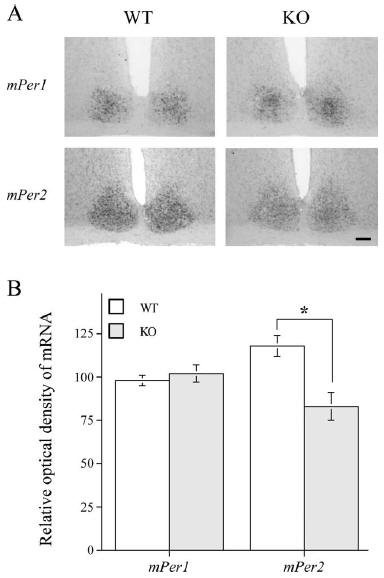
Figure 2. Representative photomicrographs (A) and quantification (B) of mPer1
and mPer2
mRNA in the SCN using in situ hybridization. Animals (n =4 per group) were given
a light pulse
(300 lux for 30 min) at CT 16 and killed 90 min after the beginning of the light
pulse. Alternate
SCN sections from each animal were processed with mPer1 and mPer2 in situ
probes, respectively.
Scale bar, 300 μm. Data are presented as mean ±SEM. *p <0.05, two-sample t test.
mPer1 and mPer2 mRNA expression after phase-delaying
light pulses
There was no significant difference in induction of mPer1 in the
SCN of DARPP-32 KO and WT mice (Fig. 2). In contrast, mPer2
staining in the SCN was reduced in the DARPP-32 KO mice (Fig.
2). In bothWTand KO strains, there were only a few cells labeled
with mPer1 or mPer2 in the SCN of control animals that were not
exposed to a light pulse, as reported previously (Yan and Silver,
2002).
DARPP-32 mRNA expression in the SCN, IGL, and retina
DARPP-32 mRNA expression was examined by in situ hybridization
in the SCN and in the retina and IGL, regions related to
photic transduction in the circadian system. In contrast to the
strong in situ signal in the striatum (Fig. 3A,B), DARPP-32 expression
was not detected in the SCN (Fig. 3A,C) or IGL (Fig.
3D) during subjective day (CT 4). We also examined animals
during nighttime and after light exposure at CT 16 but did not
detect any DARPP-32mRNAexpression in SCN or IGL (data not
shown). In contrast to the findings for the SCN, cells of the proximal
portion of the inner nuclear layer (INL) of the retina, at the
border with the inner plexiform layer, were DARPP-32 positive
(Fig. 3F).
To confirm the in situ results, we further examined DARPP-32
expression in the SCN by reverse transcription-PCR using lasercaptured
SCN tissue. The results indicate that DARPP-32 was
expressed in forebrain used as control tissue but not in the SCN
from the same PCR run (data not shown).
Discussion
The present results demonstrate that DARPP-32 KO mice displayed
attenuated phase delays to light exposure, suggesting that
their photic response is impaired. Furthermore, mice bearing a
point mutation at Thr34 (but not Ser130 mutants) have the same
deficit as do the DARPP-32 KO mice. These results suggest that
DARPP-32 is a component of the pathway mediating the photic
response and also suggest that phosphorylation of Thr34 and
activation of the downstream pathway is the responsible site of
action. In the SCN, light-induced mPer2 expression was also attenuated
in the DARPP-32 KO mice compared with that observed
in WT controls. DARPP-32 mRNA was observed in the
retina but was not detectable in the SCN or IGL. Together, our
results suggest that DARPP-32 signaling is important within the
retina, in relation to the photic input pathway to the SCN, but
does not act directly on the SCN.
The phase-advance component of the response to light was
not affected in the DARPP-32 KO mice. The phase-response
curve of C57 mice has a high amplitude of phase delay of ~1.1 h
and a low amplitude of phase advance of 0.5 h (Schwartz and
Zimmerman, 1990). The lack of difference between the WT and
DARPP-32 KO animals may be attributable to the low amplitude
of the response. Alternatively, it may be that phase delays and
advances involve different signaling pathways and clock genes
(Prosser et al., 1989; Ding et al., 1998; Albrecht et al., 2001). Our
results are consistent with the latter conclusion and suggest that
DARPP-32 participates in the pathways mediating phase delays
but not those underlying phase advances.
In DARPP-32 KO mice, we found decreased mPer2 induction
by light. This is in agreement with previous studies suggesting a
relationship between the light-induced mPer2 gene and the
phase-delay response (Albrecht et al., 2001; Yan and Silver, 2002;
Yan et al., 2003). It is unlikely that there are differences in mPer1
induction between the DARPP-32 KO and WT mice at time
points not examined here, because the peak response occurs after
90 min of light exposure for both mPer1 and mPer2 (Takumi et
al., 1998; Yan and Silver, 2002).
DARPP-32 is not directly involved in SCN clock function
It is well documented that DARPP-32 is localized in neurons
receiving dopaminergic input, with few exceptions (Ouimet et
al., 1984). Dopaminergic input is absent in the adult SCN (Ishida
et al., 2002), although dopamine D1 receptors are present in both
the fetal and adult SCN (Weaver et al., 1992; Ishida et al., 2002).
Dopaminergic regulation participates in the entrainment of the
fetal circadian clock. Periodic treatment with a dopamine D1
receptor agonist can entrain the fetuses of SCN-lesioned dams
(Viswanathan et al., 1994). Activation of D1 receptors activates
c-Fos expression in the fetalSCNbut not after postnatal day 2, the
time at which RHT innervation to the SCN occurs and photic
entrainment begins (Weaver et al., 1992, 1995). These results
indicate a transition from dopaminergic to photic regulation
during development. Together, these results suggest that dopamine
and DARPP-32 signaling do not directly regulate the clock
function within the adult SCN.
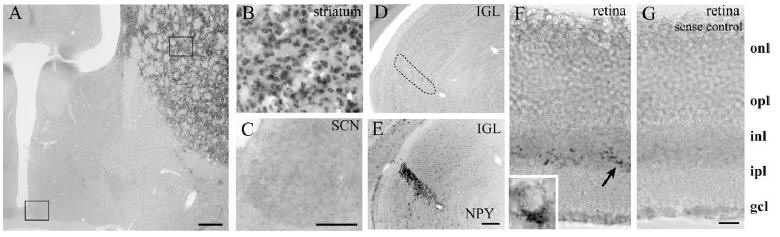
Figure 3. Expression of DARPP-32mRNAin SCN, striatum, IGL, and retina. A,
Coronal section showing robust DARPP-32 staining in the striatum and its absence
in the SCN. The box in A delineates the area shown in higher-power images of
striatum and SCN in B and C, respectively. D, DARPP-32 is
not expressed in the IGL (delineated by a dashed line). E, NPY staining
in the section adjacent to that shown in D was used to delineate the IGL.
F, DARPP-32 is expressed in the proximal portion of the inner nuclear layer.
F, Inset, High-power image of a DARPP-32-stained cell. G, When using a sense
probe, no DARPP-32 staining was seen in the retina. onl, Outer nuclear layer;
opl, outer plexiform layer; inl, inner nuclear layer; ipl, inner plexiform
layer; gcl, ganglion cell layer. Scale bars,
A, E (for D, E), 300 μm; C (for B, C), 100 μm; G
(for
F, G), 20 μm.
DARPP-32 mediates photic information in the retina
The retina sends photic information to the SCN directly through
theRHT(Moore and Lenn, 1972), which originates from a subset
of retinal ganglion cells (Gooley et al., 2001). We did not observe
DARPP-32 in these intrinsically photosensitive ganglion cells but
did find DARPP-32mRNAexpression in the proximal portion of
the INL, in which the amacrine cells are located. This is in agreement
with the finding of DARPP-32-like immunoreactivity in
the AII amacrine cells of INL in rat retina (Partida et al., 2004)
and mouse retina (P. Witkovsky and R. Silver, unpublished data).
AII amacrine cells are a component of the pathway transmitting
information from rod photoreceptors. Rod photoreceptor signals
are conveyed through rod bipolar cells to AII amacrine cells.
The output of the AII amacrine cells differentially affect ON and
OFF of cone bipolar cells, thereby altering the responses of ON
and OFF of ganglion cells (Bloomfield and Dacheux, 2001).
Melanopsin-containing photoreceptor cells in the ganglion layer
and the traditional rod and cone photoreceptor cells both contribute
to photic transduction of the circadian system (Hattar et
al., 2003; Panda et al., 2003). It has been suggested in a study using
the pseudorabies virus tract tracing method that amacrine cells in
the retina also contribute to circadian response to light (Provencio
et al., 1998).
Our results suggest a retinal site of DARPP-32 action involved
in the attenuation of phase delays seen in the KO and Thr34
mutants. However, it should be noted that DARPP-32 is widely
distributed in the brain, including cortex, thalamus, and hypothalamus
subregions, and these brain regions send projections to
various target areas (Svenningsson et al., 2004). The DARPP-32
containing cells and their targets in the brain may influence the
SCN function indirectly through either neuronal or humoral
pathways.
Significance of Thr34 phosphorylation of DARPP-32
DARPP-32 can be regulated by various neurotransmitters, neuromodulators,
and neuropeptides. Dopamine, serotonin, GABA,
adenosine, nitric oxide, and neurotensin can increase the phosphorylation
of the Thr34 site (Svenningsson et al., 2004). When
phosphorylated at Thr34, DARPP-32 is converted into an inhibitor
of PP-1 (Hemmings et al., 1984). PP-1 controls the phosphorylation
state and physiological activities of many substrates.
Thus, the neurotransmitters that increase DARPP-32 phosphorylationat T
hr34 inhibit PP-1 activity and regulate a downstream
cascade that involves receptors, ion channels, and transcription
factors (Greengard, 2001).
Dopamine signaling in retinal function
Dopamine is a chemical messenger for adaptation to light (Witkovsky,
2004). Dopaminergic interneurons are found in the retinas
of all vertebrate classes (Ehinger, 1983; Brecha et al., 1984;
Marc, 1986) and affect retinal synaptic pathways and signal processing
through dopamine receptors (Witkovsky and Sche`utte,
1991). Dopamine D1 and D2 receptors are distributed
throughout
the retina (Witkovsky, 2004). Dopamine D2 receptor KO
mice display impaired light masking but intact phase delays, supporting
a role for theD2 receptor in masking of circadian rhythms
(Doi et al., 2006). The effect of dopamine on phosphorylation of
DARPP-32 at Thr34 site is bidirectional, with dopamine D1 receptors
being stimulatory and D2 receptors inhibitory (Svenningsson
et al., 2004). Given this inhibitory action ofD2 receptors
on Thr34-DARPP-32, it is not surprising that the behavioral responses
of DARPP-32 KO and Thr34 mutants are different from
those of D2-receptor KO mice. The present study indicates an
attenuated behavioral phase-delay response in the DARPP-32
KO mice after nighttime light exposure and the expression of
DARPP-32 in the retina but not in the SCN or IGL. DARPP-32 is
found in amacrine cells (Partida et al., 2004), and AII amacrine
cells are dopamine sensitive (Witkovsky, 2004). Although dopamine
signaling has been well studied in the retina, the present
results provide the first functional evidence of a role for
DARPP-32 in the circadian response to light.


